Nitrogen is one of the most important nutrients for crop growth, second only to water, and is the major nutrient the producer can control. Nitrogen exists in many different chemical forms and passes around natural and agricultural ecosystems in a cycle. The various forms of nitrogen determine its availability to plants or whether nitrogen escapes and is no longer available to plants. The supply of useable nitrogen and the rate of losses from the soil affects the sustainability of production. Mismanaged, it can result in economic loss to the producer and have environmental repercussions, or both.
How plants use nitrogen
Nitrogen is one of the main chemical elements required for plant growth and reproduction. Nitrogen is a component of chlorophyll and therefore essential for photosynthesis. It is also the basic element of plant and animal proteins, including the genetic material DNA and RNA, and is important in periods of rapid plant growth.
Plants use nitrogen by absorbing either nitrate or ammonium ions through the roots. Most of the nitrogen is used by the plant to produce protein (in the form of enzymes) and nucleic acids. Nitrogen is readily transported through the plant from older tissue to younger tissues. Therefore, a plant deficient in nitrogen will show yellowing in the older leaves first due to the underdevelopment or destruction of chloroplasts and an absence of the green pigmented chlorophyll.
Chemical forms of nitrogen
-
Nitrogen gas (N2) makes up 78 per cent of atmosphere. It is not directly available for use by plants but is directly used in nitrogen fixation and industrial fertiliser manufacture.
-
Ammonia (NH3) is a gas that contains nitrogen. In this form the nitrogen is unavailable to plants. It is, however, involved in the cycling of nitrogen in the soil.
-
Nitrate (NO3ˉ) is an ionic form of nitrogen and is the most common form available to plants. In this form, nitrogen is mobile, leachable and usually the end product of mineralisation.
-
Ammonium (NH4+) is also an ionic form of nitrogen available to plants. Plants use less energy for uptake in this form when compared to nitrate. Nitrogen in this form is less likely to be lost from the soil than other forms.
-
Nitrite (NO2ˉ) is an intermediate in the conversion of ammonium to nitrate. Nitrogen in this form is not available to plants and is more prone to be lost from the soil than either nitate or ammonium.
-
Nitrous Oxide (N2O) is a greenhouse gas. Soil nitrogen can be lost in this form through the process of denitrification.
-
Nitric Oxide (NO) is also a gas and nitrogen in this form is lost through denitrification. It may be harmful to the ozone layer.
All of these above forms of nitrogen are known as inorganic forms of nitrogen.
Organic nitrogen compounds are complex and unavailable to plants. They are the end products of immobilisation.
The nitrogen cycle
Below is diagram showing the nitrogen cycle, the various forms of nitrogen, the processes that convert one form of nitrogen to another and the part of the environment in which these processes occur. There are many variations of this basic nitrogen cycle diagram.
Nitrogen may be found in air, water or land; nitrogen may exist in several different chemical forms; it will undergo many changes in form and location throughout the cycle due to processes which occur as a result of weather, plants, animals and humans. In some cases nitrogen may be lost to the soil nitrogen cycle. It is important to determine whether human activity has accelerated the rate of natural losses and whether these losses pose an environmental threat.
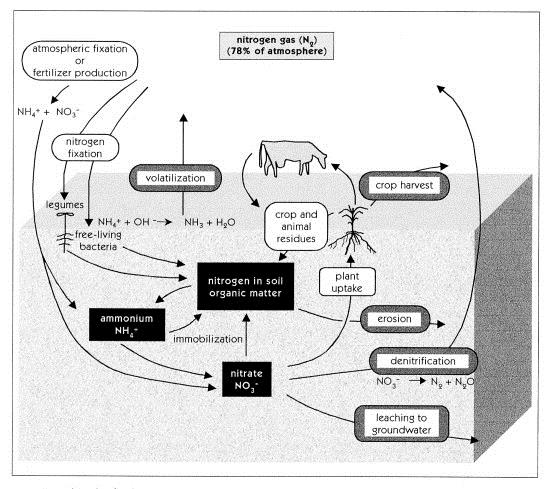
Nitrogen cycle

